|
Visit Bodycote’s new Interactive Periodic Table to view this information and more graphically. Chemical symbolįrom the Latin Argentum which originally meant shining metalįrom the Latin Aurum which originally meant yellowįrom Cuprum, the Latin name for Cyprus, the Roman source of copperįrom the Latin Hydragyrummeaning liquid silverįrom Natrium, the Latin namefor sodium carbonateįrom the Latin Plumbummeaning soft white metalįrom the Latin Stibiummeaning cosmetic powderįrom Wolfram, an old name for the element derived from its ore, Wolframite In those cases, the origin of the symbol used is given. A very few elements have symbols which appear to have no relationship with their names. In some cases, the first letter together with some other letter from their name was used, particularly when their first two letters had already been allocated to another element. Most chemical elements are represented symbolically by two letters, generally the first two in their name. Powdermet® Selective surface net shape (SSNS)Īnti-Slavery and Human Trafficking Statement Sub-critical annealing / intercritical annealing Specialty Stainless Steel Processes (S 3P) answer is 2 times the above empirical formula, so the molecular formula is C2H4O2. Precipitation hardening: Stainless steels Determine identity of an element from a binary formula and mass data. The sign of the charges in the subscript is ignored while writing the molecular formula.Case hardening with subsequent hardening operationĬase hardening without subsequent hardening operationįluidised bed/salt bath nitriding/nitrocarburising However, one Ca 2+ cation requires 2 Cl atoms as 2Cl - to balance the charges and form a neutral compound.īy criss-crossing the numerical value of charges and using it as a subscript, the correct ratio of the ions is obtained to conclude the formula of AB 2 as CaCl 2. Moreover, elements could repeat in the vector C and this fact is lost when we. The valency of Cl is 1, and one Cl atom can accept one electron to form one Cl - anion. (a) Chlorine and cesium (b) Calcium and astatine. The valency of Ca is 1, and the one Ca atom comfortably loses two electrons to form Ca 2+ cation. Write Lewis symbols for the elements both before and after chemical combination. The simplest method is to employ the criss-cross method. Criss-cross (cross-multiplication) method to deduce the Molecular Formula One Ca atom, and two Cl atoms form the compound Calcium Chloride (CaCl 2). One Cl atom can only pick one electron therefore, the two electrons are accepted by 2 Cl atoms to give 2Cl - anions. 
The Group 2 Calcium (Ca) atom can easily lose two electrons, therefore, The element B from the Group 7 and period 3 is Chlorine (Cl). The principal quantum number of the outermost valence shell of element B is 3 therefore, the element belongs to the 3 rd period (row) of the periodic table. Therefore, halogens have high negative electron gain enthalpy values and attain electrons easily. 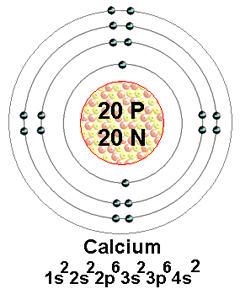
The halogens are less by one electron from attaining the stable inert gas configuration. 
The principal quantum number of the outermost shell of element A is 4 therefore, the element belongs to the 4 th period (row) of the periodic table.Įlement A belonging to Group 2 and period 4, is Calcium (Ca).ī belongs to group 7 based on the valence shell electronic configuration of 7. Group 2 elements can therefore lose two electrons to attain stability. Metals have low ionization enthalpy they can easily lose electrons to attain the nearest stable, inert gas configuration. Therefore, A belongs to the Group 2 of the periodic table. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. What is the formula of the ionic compound formed between these elements?Ī has a valence shell configuration of 2. Calcium is a chemical element with the symbol Ca and atomic number 20. The electronic configuration of two elements A and B are.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
